By Tags

How to Modify Dose Descriptors to A Correct Starting Point
2016-08-10
When calculating derived no-effect level (DNEL), it is very important to convert certain&n Read More.

How to Calculate Predicted No-Effect Concentration (PNEC)
2016-07-13
Predicted No-Effect Concentration (PNEC) is the concentration of a substance in any e Read More.

Definition of Toxicological Dose Descriptors (LD50, LC50, EC50, NOAEL, LOAEL, etc)
2016-06-17
In toxicology and eco-toxicology, dose descriptor is the term used to identify the relatio Read More.
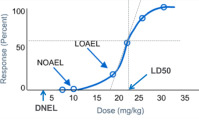
How to Derive Derived No-Effect Level (DNEL)
2016-06-14
Derived No-Effect Level (DNEL) is defined as the level of chemical exposure above whi Read More.

Chemical Risk Assessment: Overview and Examples
2016-05-17
In this article, we will give you an introduction to chemical risk assessment. We will foc Read More.

Chemical Risk Assessment: Ecotoxicology and Environmental Fate
2016-04-27
The following ecotoxicology and environmental fate studies are often used for GHS hazard Read More.

Chemical Risk Assessment: Toxicology
2016-04-27
Toxicology is defined as the study of the adverse effects of chemicals or physical agents Read More.

Toxicokinetics
2016-04-26
Toxicokinetics(TK) is essentially the study of how a substance gets into the body and Read More.

Carcinogenicity
2016-04-26
A carcinogen is a chemical substance or a mixture of chemical substances that induces canc Read More.

Reproductive and Developmental Toxicity
2016-04-25
Reproductive toxicity is defined as adverse effects of a chemical substance on sexual func Read More.

Repeated Dose Toxicity
2016-04-25
The primary goal of repeated dose toxicity studies is to characterize the adverse toxicolo Read More.

Mutagenicity and Genotoxicity
2016-04-24
Mutagenicity refers to the induction of permanent transmissible changes in the amount or s Read More.