GHS Classification Criteria for Reproductive Toxicity
Little Pro on 2018-04-09
Reproductive toxicity refers to adverse effects of a chemical substance/mixture on sexual function and fertility in adult males and females, as well as developmental toxicity in the offspring. Developmental toxicity pertains to adverse toxic effects to the developing embryo or fetus.
GHS Classification Criteria for Reproductive Toxicity
There are 3 hazard categories for reproductive toxicity under GHS.
| Category | Criteria |
|---|---|
| Category 1A | Known human reproductive toxicants
Based on evidence from humans. |
| Category 1B | Presumed human reproductive toxicants - largely based on animal studies.
|
| Category 2 | Suspected human reproductive toxicant - Evidence from animal and/or human studies is limited
|
| Effects on via lactation | Effects on via lactation Toxicants which may interfere with lactation or which may be present in breast milk and may cause harm to breast-fed children |
Testing Guidelines for Reproductive/Developmental Toxicity
The following studies can provide evidence for GHS classification.
- Reproductive/Developmental Toxicity Screening Assay (OECD TG 421)
- Prenatal developmental toxicity test (OECD TG 414)
- Two-generation reproduction toxicity study (OECD TG 416)
- Extended one-generation reproductive toxicity study (EOGRTS; OECD TG 443)
Reproductive Toxicity Classification for Mixture
The picture below shows you the concentration limit of a reproductive toxicant triggering the reproductive toxicity classification of a mixture.
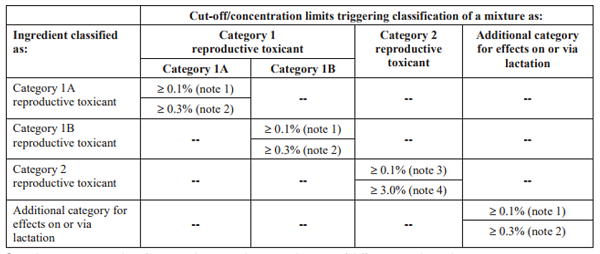
Note: Different authorities may have adopted different concentration limits. More info can be found below.
References and More Reading
- GHS classification criteria in a single page
- UN GHS purple book - health hazards
- GHS classification criteria - acute toxicity
- GHS classification criteria - skin irritation and corrosion
- GHS classification criteria - eye irritation and corrosion
- GHS classification criteria - skin sensitization
- GHS classification criteria - specific target organ toxicity (STOT)
- GHS classification criteria - germ cell mutagenicity
- GHS classification criteria - reproductive toxicity
- GHS classification criteria - carcinogenicity
- M-factor and GHS Classification Criteria for Aquatic Environment
Having Questions?
We do not provide consultancy services. If you have questions or need any help, please contact our sponsor. You may also find an expert in CSP business directory below. If you are a consultant, you may get yourself listed in CSP business directory (free) or sponsor this page to leave your contact info on this page..

Tags: Topics - GHS, GHS Classification