How to Use ECOSAR to Predict Aquatic Toxicity
Little Pro on 2017-10-13
Ecological Structure Activity Relationships Program (ECOSAR) is a frequently used QSAR tool developed by US EPA to predict a chemical's acute (short-term) toxicity and chronic (long-term or delayed) toxicity to aquatic organisms, such as fish, aquatic invertebrates, and aquatic plants. In this tutorial, we will show you how to use ECOSAR and how to interpret ECOSAR's output result. In the end, we will talk about its limitations.
How to Install and Use ECOSAR
ECOSAR is a free tool, easy to install and use. It can be downloaded here. After you have downloaded it and installed it, you will see the following main screen. To start using ECOSAR, you only need to input very limited substance info and then click "calculate" button.
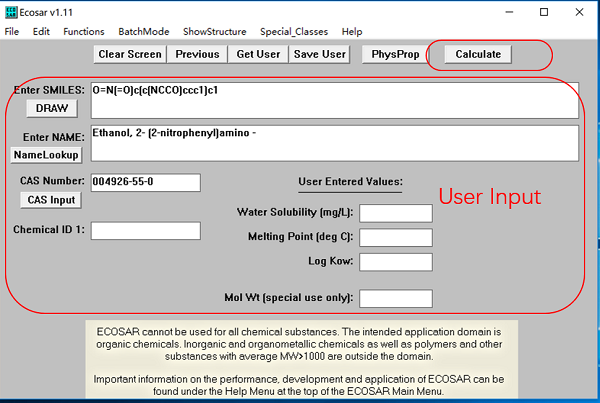
User Inputs for ECOSAR
To run an ECOSAR estimation, a user may input the following info:
- SMILES notation: It is a molecular text code of displayed chemical STRUCTURE. You can search it in ECOSAR's SMILES database by CAS no., molecular structure or input it directly. This field is required.
- Chemical Name: This field is not required but recommended.
- CAS Number: This field is optional; not required.
- User-entered Log Kow: This field is optional; not required.
- User-entered Water Solubility: The user-entered water solubility value in mg/L. This field is optional; not required.
- User-entered Melting Point: The user-entered melting point in degrees Celsius (ºC). This field is optional; not required.
After a SMILES notation has been entered, the ECOSAR program will choose appropriate ECOSAR class(es) (i.e, neutral organics, esters, acrylates) from the SMILES notation and calculate the ecotoxicity QSARs using a log Kow value. If measured LogKow, water solubility, and melting point has not been entered, the ECOSAR will use results estimated by EPI Suite.
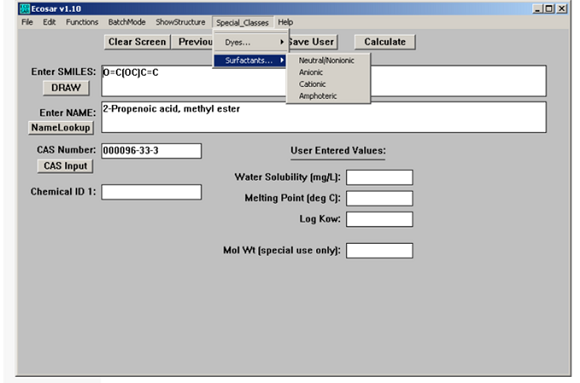
For special classes such as dyes and surfactants, you need to use the following data entry window.
What Is SMILES and How to Find It
SMILES (Simplified Molecular Input Line Entry System) is a molecular text code of displayed chemical STRUCTURE. It is required as input by many QSAR models. ECOSAR has incorporated a SMILES database in which you can search for a SMILES by CAS no or chemical name.
For a big majority of chemical substances, you may find SMILES via the links below.
An online SMILES translator is available from the National Cancer Institute at http://cactus.nci.nih.gov/services/translate/. For a brand new chemical substance, you usually need to draw its chemical structure using commercial software. The software will then give you the SMILES code.
How to Interpret ECOSAR Output Result
We will use Ethanol, 2-((2-nitrophenyl)amino)- as an example.
- Chemical name: Ethanol, 2-((2-nitrophenyl)amino)-
- CAS no.: 4926-55-0
- Smiles: O=[N+]([O-])
c1c(NCCO) cccc1
After you have entered above info to ECOSAR and clicked "calculate" button, you will get the following ECOSAR output result. The result includes chemical attributes, whether measured data is available, chosen ECOSAR Class, estimated aquatic toxicity values, baseline toxicity values, and information specific to the interpretation of the QSAR results.
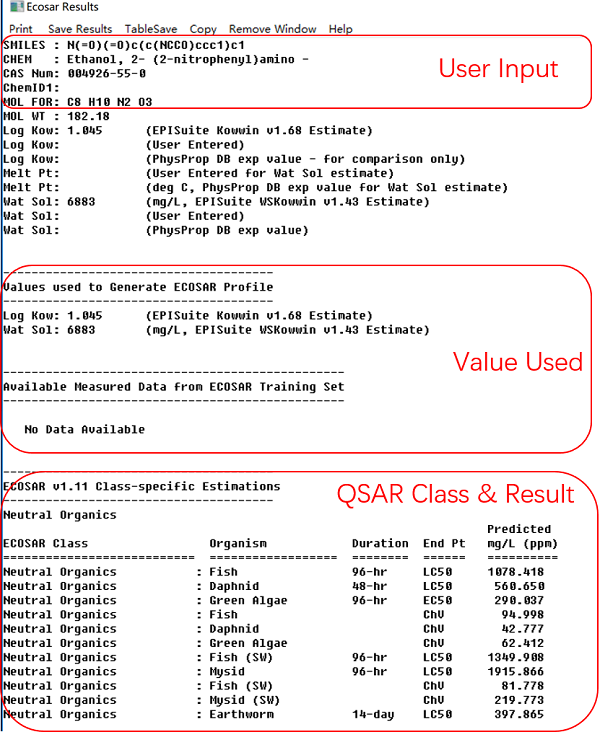
Acute aquatic toxicity estimate is given in LC50 or EC50 (mg/L) depending on species (fish, daphnid or algae). Lower value indicates higher toxicity.
Chronic aquatic toxicity estimate is expressed in ChV (in mg/L). The ChV, or Chronic Value, is defined as the geometric mean of the no observed effect concentration (NOEC) and the lowest observed effect concentration (LOEC). The Concentration of Concern (COC) is the lowest ChV divided by an uncertainty factor (assessment or safety factor) of 10.
More info about LC50, EC50, LOEC, and NOEC can be found here.
Choice of Representative QSAR Class and Value
For a complex chemical substance with multiple functional groups (esters, ketones), multiple QSAR classes may be chosen by ECOSAR. Then you will get different LC50, EC50 and ChVs (see example below) for each QSAR class. Unless you have solid evidence to choose the most representative QSAR class, the lowest estimated values shall be used for further assessment. In this example, aquatic toxicity estimates from pyrethroids class will be used.
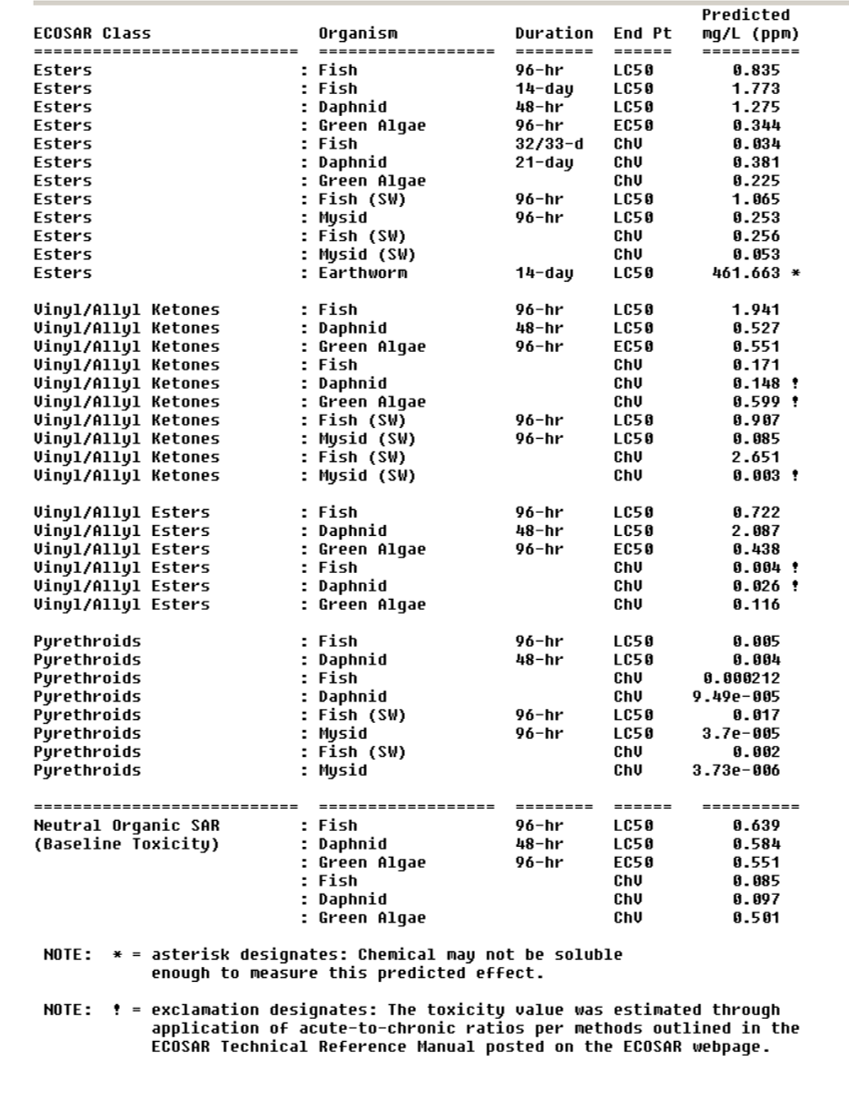
Baseline Toxicity and Mode of Action
In above example, you can also see the value of baseline toxicity given. Basline toxicity is related to the mode of action of a chemical substance. Many types of chemical classes mainly present toxicity to organisms via narcosis (i.e , neutral organics, ethers, alcohols, ketones). However, some organic chemical classes have been identified as having a more specific mode of toxicity. These are typically organics that are reactive and/or ionizable and exhibit excess toxicity in addition to narcosis (i.e. acrylates, epoxides, anilines).The purpose for presenting the baseline toxicity values is so the user can quantify the amount of excess toxicity above baseline narcosis for the chemical class, if interested.
Limitations of ECOSAR
1. ECOSAR can only be used to evaluate organic chemical substances with discrete structures. It does not work for inorganic or organometallic chemicals, polymers and chemicals with MW >1,000, mixtures and nanomaterials.
2. Estimated aquatic toxicity values given by ECOSAR can only be used for screening purpose and determining whether further aquatic toxicity testing is necessary. They can also be used for determining more sensitive species. However, the estimated values cannot be used for quantitative environmental risk assessment.
3. There is log Kow cut-off for ECOSAR to work. In general, when the log Kow is ≤ 5.0 for fish and daphnid, or ≤ 6.4 for green algae, ECOSAR provides reliable estimates for acute effects. If the log Kow exceeds those limits, empirical data indicate that the decreased solubility of these lipophilic chemicals results in “no effects at saturation” during a 48- hour to 96-hour test. For chronic exposures, the log Kow cut off is 8.0 or greater (indicating a poorly soluble chemical) and “no effects at saturation” are expected in saturated solutions even with long-term exposures.
4. For solid chemical substances, it is important to compare the toxicity estimates with the water solubility.
Reference
Having Questions?
We do not provide consultancy services. If you have questions or need any help, please contact our sponsor. You may also find an expert in CSP business directory below. If you are a consultant, you may get yourself listed in CSP business directory (free) or sponsor this page to leave your contact info on this page..

Tags: Topics - CRA, Chemical Risk Assessment Tutorials